Abstract
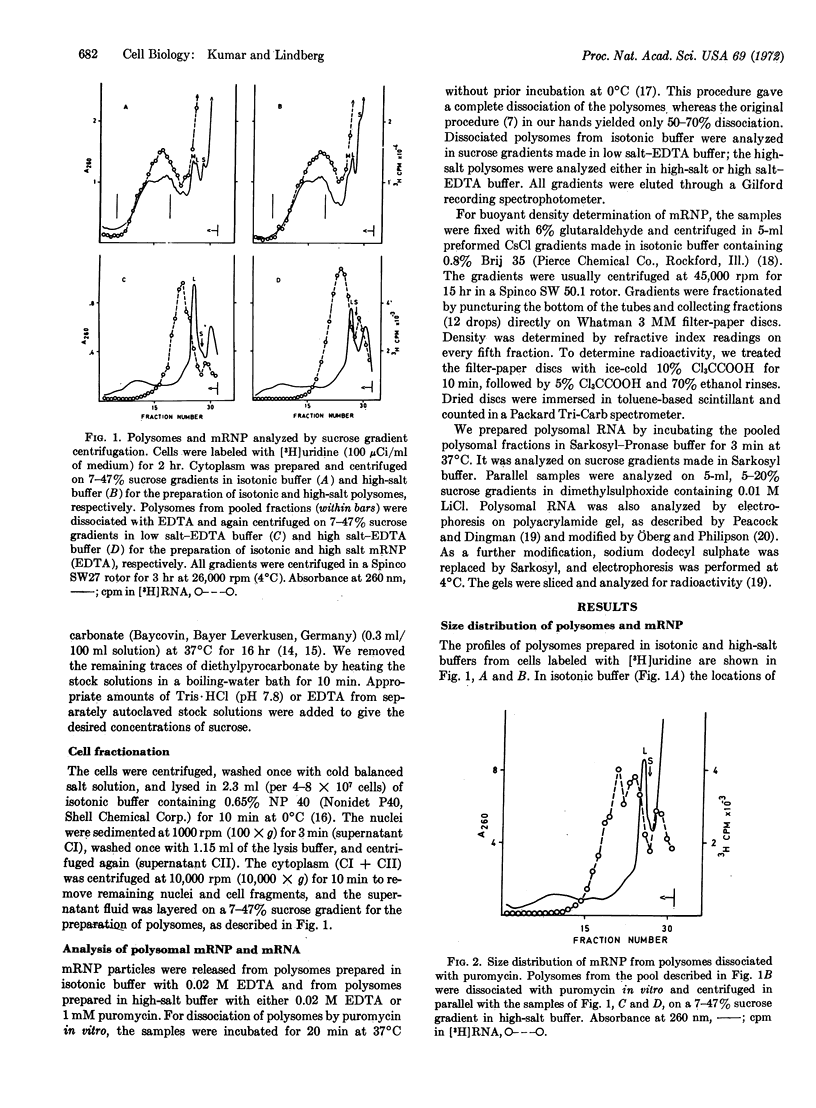
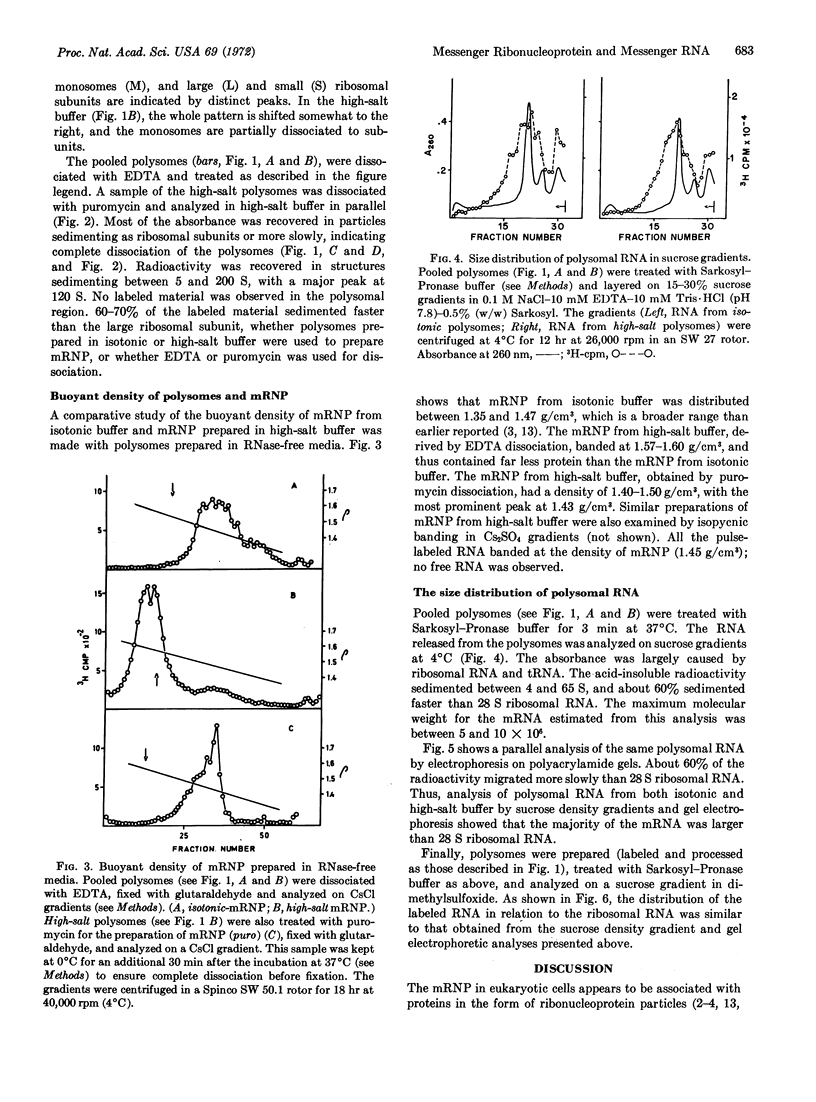
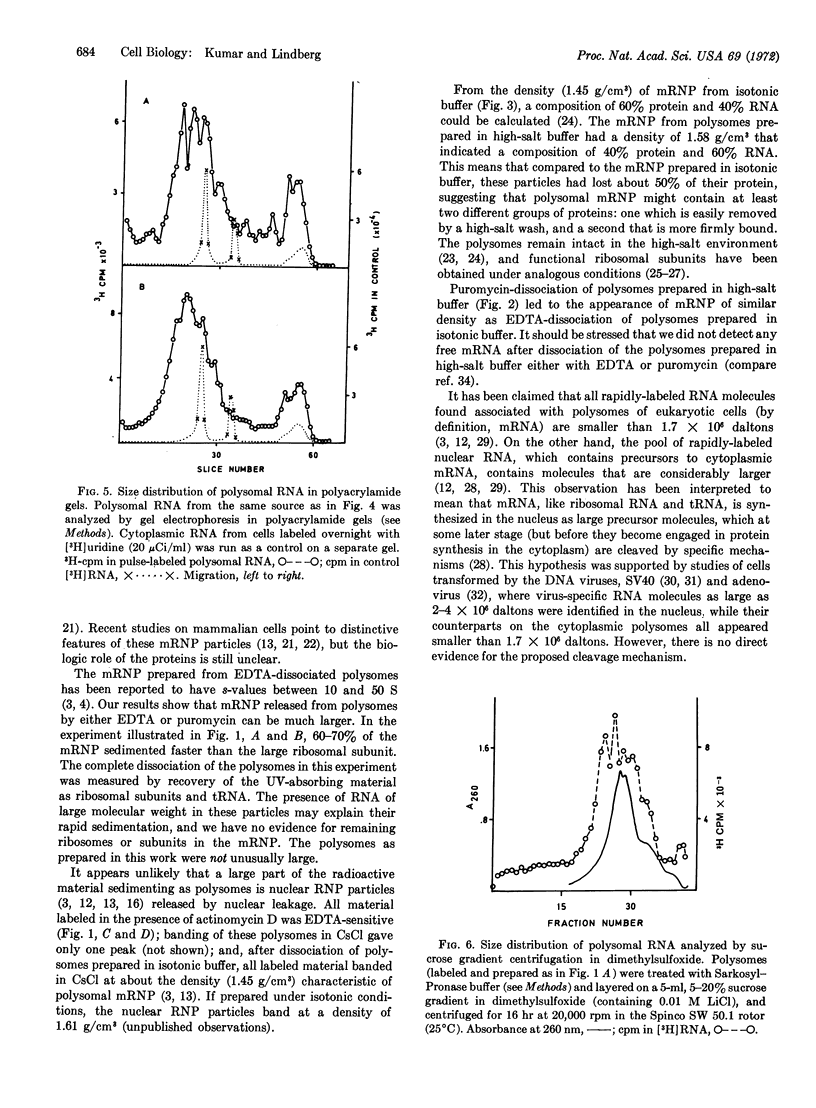
Messenger ribonucleoprotein and mRNA from KB-cells were isolated under conditions designed to minimize nonspecific RNA-protein interaction and to minimize degradation by contaminating ribonucleases. A large fraction, 60-70%, of the messenger ribonucleoprotein from polysomes dissociated in vitro by either EDTA or puromycin sedimented faster than the large ribosome subunit. Messenger ribonucleoprotein particles with sedimentation coefficients up to 200 S were observed. Released mRNA was also large, with maximal molecular weights around 5 × 106.
Keywords: eukaryotic cells, polysomes, molecular weight, density gradient centrifugation
Full text
PDF

Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Baltimore D., Huang A. S. Interaction of HeLa cell proteins with RNA. J Mol Biol. 1970 Feb 14;47(3):263–273. doi: 10.1016/0022-2836(70)90301-3. [DOI] [PubMed] [Google Scholar]
- Baltimore D., Huang A. S. Isopycnic separation of subcellular components from poliovirus-infected and normal HeLa cells. Science. 1968 Nov 1;162(3853):572–574. doi: 10.1126/science.162.3853.572. [DOI] [PubMed] [Google Scholar]
- Blobel G. Release, identification, and isolation of messenger RNA from mammalian ribosomes. Proc Natl Acad Sci U S A. 1971 Apr;68(4):832–835. doi: 10.1073/pnas.68.4.832. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Blobel G., Sabatini D. Dissociation of mammalian polyribosomes into subunits by puromycin. Proc Natl Acad Sci U S A. 1971 Feb;68(2):390–394. doi: 10.1073/pnas.68.2.390. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Borun T. W., Scharff M. D., Robbins E. Preparation of mammalian polyribosomes with the detergent Nonidet P-40. Biochim Biophys Acta. 1967 Nov 21;149(1):302–304. doi: 10.1016/0005-2787(67)90715-0. [DOI] [PubMed] [Google Scholar]
- Darnell J. E., Jr Ribonucleic acids from animal cells. Bacteriol Rev. 1968 Sep;32(3):262–290. doi: 10.1128/br.32.3.262-290.1968. [DOI] [PMC free article] [PubMed] [Google Scholar]
- EAGLE H. Amino acid metabolism in mammalian cell cultures. Science. 1959 Aug 21;130(3373):432–437. doi: 10.1126/science.130.3373.432. [DOI] [PubMed] [Google Scholar]
- Falvey A. K., Staehelin T. Structure and function of mammalian ribosomes. I. Isolation and characterization of active liver ribosomal subunits. J Mol Biol. 1970 Oct 14;53(1):1–19. doi: 10.1016/0022-2836(70)90042-2. [DOI] [PubMed] [Google Scholar]
- Girard M., Baltimore D. The effect of HeLa cell cytoplasm on the rate of sedimentation of RNA. Proc Natl Acad Sci U S A. 1966 Sep;56(3):999–1002. doi: 10.1073/pnas.56.3.999. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Henshaw E. C. Messenger RNA in rat liver polyribosomes: evidence that it exists as ribonucleoprotein particles. J Mol Biol. 1968 Sep 28;36(3):401–411. doi: 10.1016/0022-2836(68)90164-2. [DOI] [PubMed] [Google Scholar]
- Lebleu B., Marbaix G., Huez G., Temmerman J., Burny A., Chantrenne H. Characterization of the messenger ribonucleoprotein released from reticulocyte polyribosomes by EDTA treatment. Eur J Biochem. 1971 Mar 11;19(2):264–269. doi: 10.1111/j.1432-1033.1971.tb01313.x. [DOI] [PubMed] [Google Scholar]
- Lindberg U., Darnell J. E. SV40-specific RNA in the nucleus and polyribosomes of transformed cells. Proc Natl Acad Sci U S A. 1970 Apr;65(4):1089–1096. doi: 10.1073/pnas.65.4.1089. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Martin T. E., Rolleston F. S., Low R. B., Wool I. G. Dissociation and reassociation of skeletal muscle ribosomes. J Mol Biol. 1969 Jul 14;43(1):135–149. doi: 10.1016/0022-2836(69)90084-9. [DOI] [PubMed] [Google Scholar]
- Martin T. E., Wool I. G. Formation of active hybrids from subunits of muscle ribosomes from normal and diabetic rats. Proc Natl Acad Sci U S A. 1968 Jun;60(2):569–574. doi: 10.1073/pnas.60.2.569. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Monneron A., Liew C. C., Allfrey V. G. Isolation and biological activity of mammalian helical polyribosomes. J Mol Biol. 1971 Apr 28;57(2):335–350. doi: 10.1016/0022-2836(71)90350-0. [DOI] [PubMed] [Google Scholar]
- Oberg B., Philipson L. Replication of poliovirus RNA studied by gel filtration and electrophoresis. Eur J Biochem. 1969 Dec;11(2):305–315. doi: 10.1111/j.1432-1033.1969.tb00774.x. [DOI] [PubMed] [Google Scholar]
- Peacock A. C., Dingman C. W. Molecular weight estimation and separation of ribonucleic acid by electrophoresis in agarose-acrylamide composite gels. Biochemistry. 1968 Feb;7(2):668–674. doi: 10.1021/bi00842a023. [DOI] [PubMed] [Google Scholar]
- Penman S., Vesco C., Penman M. Localization and kinetics of formation of nuclear heterodisperse RNA, cytoplasmic heterodisperse RNA and polyribosome-associated messenger RNA in HeLa cells. J Mol Biol. 1968 May 28;34(1):49–60. doi: 10.1016/0022-2836(68)90234-9. [DOI] [PubMed] [Google Scholar]
- Perry R. P., Kelley D. E. Buoyant densities of cytoplasmic ribonucleoprotein particles of mammalian cells: distinctive character of ribosome subunits and the rapidly labeled components. J Mol Biol. 1966 Apr;16(2):255–268. doi: 10.1016/s0022-2836(66)80171-7. [DOI] [PubMed] [Google Scholar]
- Perry R. P., Kelley D. E. Messenger RNA-protein complexes and newly synthesized ribosomal subunits: analysis of free particles and components of polyribosomes. J Mol Biol. 1968 Jul 14;35(1):37–59. doi: 10.1016/s0022-2836(68)80035-x. [DOI] [PubMed] [Google Scholar]
- Roberts W. K., Newman J. F. Use of low concentrations of actinomycin D in the study of RNA synthesis in Ehrlich ascites cells. J Mol Biol. 1966 Sep;20(1):63–73. doi: 10.1016/0022-2836(66)90117-3. [DOI] [PubMed] [Google Scholar]
- Solymosy F., Fedorcsák I., Gulyás A., Farkas G. L., Ehrenberg L. A new method based on the use of diethyl pyrocarbonate as a nuclease inhibitor for the extraction of undegraded nucleic acid from plant tissues. Eur J Biochem. 1968 Sep 24;5(4):520–527. doi: 10.1111/j.1432-1033.1968.tb00401.x. [DOI] [PubMed] [Google Scholar]
- Spirin A. S., Nemer M. Messenger RNA in early sea-urchin embryos: cytoplasmic particles. Science. 1965 Oct 8;150(3693):214–217. doi: 10.1126/science.150.3693.214. [DOI] [PubMed] [Google Scholar]
- Spohr G., Granboulan N., Morel C., Scherrer K. Messenger RNA in HeLa cells: an investigation of free and polyribosome-bound cytoplasmic messenger ribonucleoprotein particles by kinetic labelling and electron microscopy. Eur J Biochem. 1970 Dec;17(2):296–318. doi: 10.1111/j.1432-1033.1970.tb01168.x. [DOI] [PubMed] [Google Scholar]
- Strauss J. H., Jr, Kelly R. B., Sinsheimer R. L. Denaturation of RNA with dimethyl sulfoxide. Biopolymers. 1968 Jun;6(6):793–807. doi: 10.1002/bip.1968.360060604. [DOI] [PubMed] [Google Scholar]
- Warner J. R., Soeiro R., Birnboim H. C., Girard M., Darnell J. E. Rapidly labeled HeLa cell nuclear RNA. I. Identification by zone sedimentation of a heterogeneous fraction separate from ribosomal precursor RNA. J Mol Biol. 1966 Aug;19(2):349–361. doi: 10.1016/s0022-2836(66)80009-8. [DOI] [PubMed] [Google Scholar]
- Zylber E. A., Penman S. The effect of high ionic strength on monomers, polyribosomes, and puromycin-treated polyribosomes. Biochim Biophys Acta. 1970 Mar 19;204(1):221–229. doi: 10.1016/0005-2787(70)90505-8. [DOI] [PubMed] [Google Scholar]


